Utah health experts believe younger kids could get the Pfizer vaccine around Halloween
Sep 20, 2021, 2:06 PM | Updated: 4:10 pm
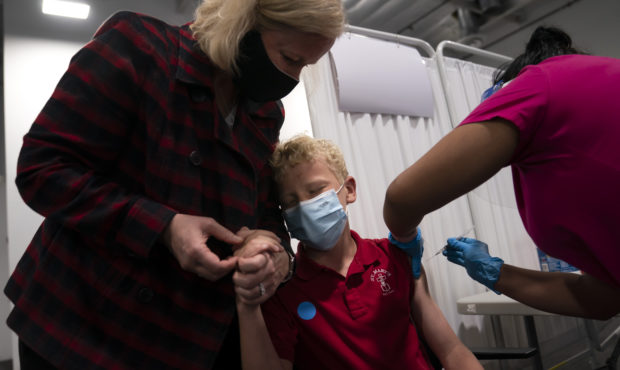
FILE -- Heather Haworth, left, holds the hand of her 12-year-old son Jeremy as he receives the first dose of the Pfizer COVID-19 vaccine from medical assistant Gloria Urgell at Providence Edwards Lifesciences vaccination site in Santa Ana, Calif., Thursday, May 13, 2021. (AP Photo/Jae C. Hong)
(AP Photo/Jae C. Hong)
Pfizer says the early results of their Phase2/3 study show that their vaccine for Covid-19 is safe for children between ages 5 and 11.
It said the two-dose vaccine is “safe, well-tolerated and showed robust neutralizing antibody responses” in children.
The study still needs to be peer-reviewed, then given to the FDA.
“Since July, pediatric cases of COVID-19 have risen by about 240 percent in the U.S. — underscoring the public health need for vaccination. These trial results provide a strong foundation for seeking authorization of our vaccine for children 5 to 11 years old, and we plan to submit them to the FDA and other regulators with urgency,” Albert Bourla, chairman and chief executive officer of Pfizer, said in a statement.
Earlier this month, both the acting FDA Commissioner and the director of FDA’s Center for Biologics Research and Evaluation, said the FDA would review data for a vaccine for younger children “as quickly as possible, likely in a matter of weeks rather than months,” once it was submitted for authorization.
“What we are hearing is the emergency use authorization could be around the end of October. The FDA has to give authorization, then the CDC gives us guidance on how exactly to give it,” said Dr. Tamara Sheffield, Intermountain Healthcare medical director with community health and prevention.
Sheffield spoke on KSL’s Dave and Dujanovic Show about the timeline.
“We just have to make sure everyone is educated and knows how to do this. It should be end of October, beginning of November that it goes to your doctor’s office or your pharmacy to give to kids.”
The Utah Department of Health says they will be ready to distribute those doses when it’s time. They are waiting like everyone else for the go-ahead, and the recommendations and authorizations.
Sheffield said the younger kids will be given two doses, but each will be a third of the size of dose that’s given to people ages 12 and up.
“Young immune systems are great immune systems to get vaccinated because they respond and give the protection needed, but you don’t have to give as high a dose to get that,” she said.
She said a lower dose should also mean fewer side effects like fever, aches, and chills.
For those younger than five, Sheffield said vaccine approvals probably won’t come for several more months.
How To Prevent the Spread of COVID-19 Coronavirus
COVID-19 coronaviruses transmitted from person to person. It is a virus that is similar to the common cold and the flu. So, to prevent it from spreading:
- Wash hands frequently and thoroughly, with soap and water, for at least 20 seconds.
- Wear a mask.
- Don’t touch your face.
- Keep children and those with compromised immune systems away from someone who is coughing or sneezing (in this instance, at least six feet)
- If there is an outbreak near you, practice social distancing (stay at home, instead of going to the movies, sports events, or other activities.)
- Get a flu shot.
- Get vaccinated.
Local resources
Utah’s Coronavirus Information
The Church of Jesus Christ of Latter-day Saints
Utah Coronavirus Information Line – 1-800-456-7707
National Resources
Centers for Disease Control and Prevention
Commonly asked questions, World Health Organization













