Pfizer/BioNTech submit initial data on Covid-19 vaccine for people ages 5 to 11 to FDA, but aren’t seeking EUA yet
Sep 28, 2021, 7:10 AM | Updated: 7:11 am
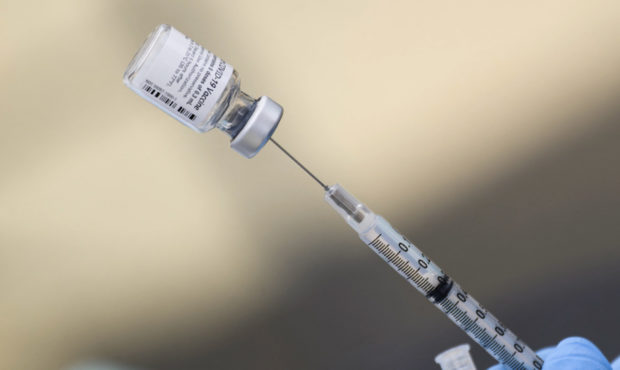
(CNN) — Pfizer and BioNTech said Tuesday they have submitted Covid-19 vaccine data on children ages 5 to 11 to the US Food and Drug Administration for initial review, but are not yet seeking emergency use authorization.
A formal submission to request EUA for the vaccine is expected to follow in the coming weeks, the companies said in a statement.
Submissions to the European Medicines Agency and other regulatory authorities are also planned, they said.
This is the first submission of data to the FDA for a Covid-19 vaccine for younger children. The Pfizer/BioNTech vaccine is approved for people age 16 and older and has an EUA for people ages 12 to 15.
Last week, Pfizer released details of a Phase 2/3 trial that showed its Covid-19 vaccine was safe and generated a “robust” antibody response in children ages 5 to 11. The trial included 2,268 participants ages 5 to 11 and used a two-dose regimen of the vaccine administered 21 days apart. This trial used a 10-microgram dose — smaller than the 30-microgram dose that has been used for those 12 and older.
Participants’ immune responses were measured by looking at neutralizing antibody levels in their blood and comparing those levels to a control group of 16- to 25-year-olds who were given a two-dose regimen with the larger 30-microgram dose. Pfizer said the levels compared well with older people who received the larger dose, demonstrating a “strong immune response in this cohort of children one month after the second dose.”
Pfizer and BioNTech said Tuesday they plan to submit data from the full Phase 3 trial for scientific peer-reviewed publication.
FDA officials have said that once vaccine data was submitted, the agency could authorize a vaccine for younger children in a matter of weeks — not months — but it would depend on the timing and quality of the data provided.
The FDA has moved quickly in the past to extend vaccine authorization to additional groups. For example, Pfizer sought emergency use authorization for its Covid-19 vaccine for people ages 12 to 15 on April 9 and the EUA was granted May 10.
However, for these younger children, FDA is expected to convene is vaccine advisers to make a recommendation on the vaccine before authorization. If the FDA OKs it, a panel of CDC vaccine advisers will meet to consider whether to recommend its use.
Following the announcement from Pfizer and BioNTech on Tuesday, Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said hopefully vaccines can go into arms by the end of October.
“The FDA takes very seriously the importance of getting vaccines shown to be safe and effective in children to getting it into the arms of children, in this case children from eleven down to five,” Fauci said on MSNBC’s Morning Joe Tuesday. “The FDA, you never want to get ahead of their judgement, but I would imagine in the next few weeks, they will examine that data and hopefully they’ll give the OK so that we could start vaccinating children, hopefully before the end of October.”
Data on the immune response and safety for two more age groups — children ages 2 to 5 and 6 months to 2 years — are expected “as soon as the fourth quarter of this year,” Pfizer and BioNTech said Tuesday.
The-CNN-Wire
™ & © 2021 Cable News Network, Inc., a WarnerMedia Company. All rights reserved.
How To Prevent the Spread of COVID-19 Coronavirus
COVID-19 coronaviruses transmitted from person to person. It is a virus that is similar to the common cold and the flu. So, to prevent it from spreading:
- Wash hands frequently and thoroughly, with soap and water, for at least 20 seconds.
- Wear a mask.
- Don’t touch your face.
- Keep children and those with compromised immune systems away from someone who is coughing or sneezing (in this instance, at least six feet)
- If there is an outbreak near you, practice social distancing (stay at home, instead of going to the movies, sports events, or other activities.)
- Get a flu shot.
- Get vaccinated.
Local resources
Utah’s Coronavirus Information
The Church of Jesus Christ of Latter-day Saints
Utah Coronavirus Information Line – 1-800-456-7707
National Resources
Centers for Disease Control and Prevention
Commonly asked questions, World Health Organization













