FDA postpones advisory panel meeting on Pfizer COVID vaccine for young kids
Feb 11, 2022, 12:09 PM
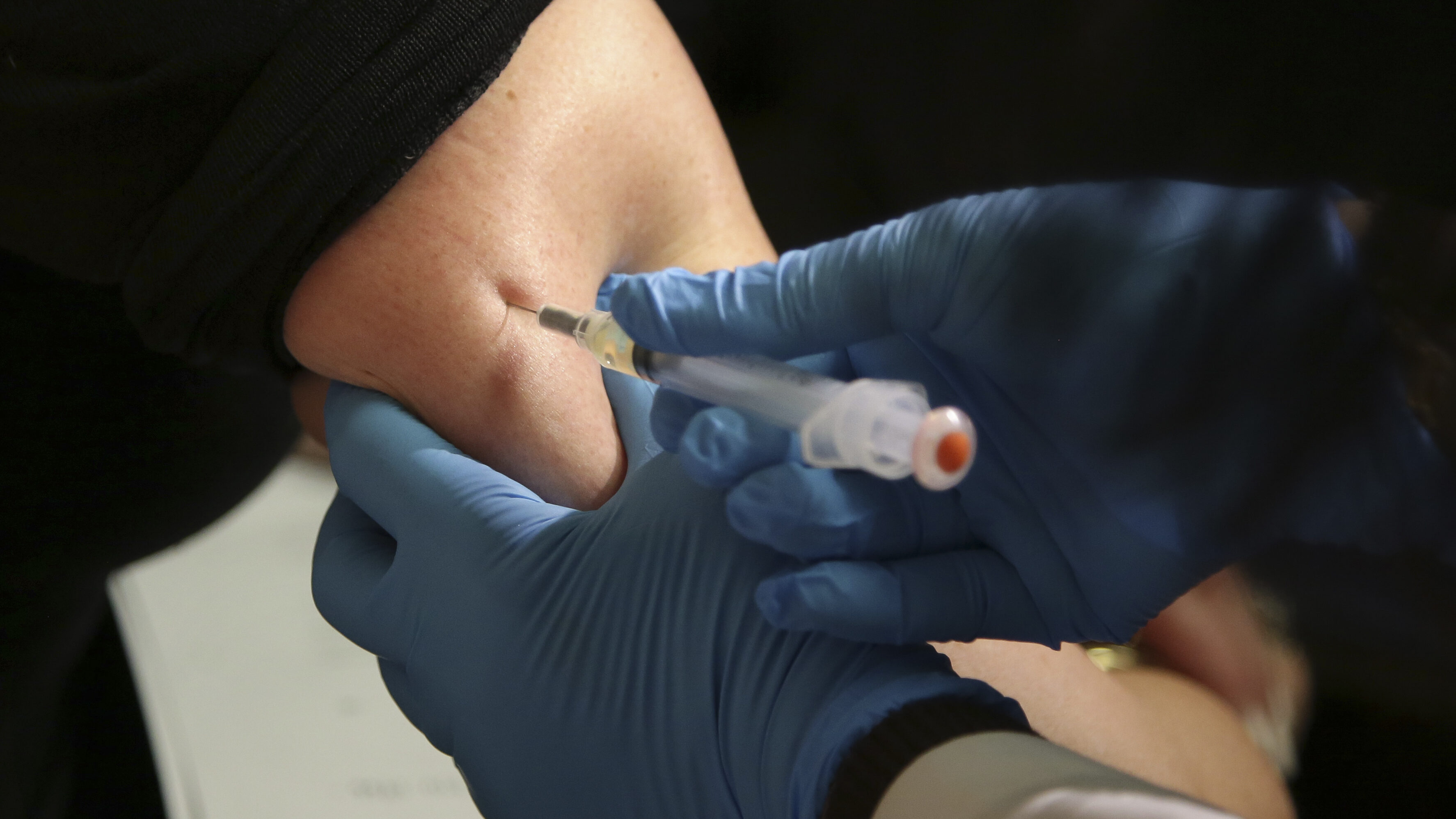
FILE - Nurse Lydia Holly prepares a child's COVID-19 vaccine dose, on Nov. 3, 2021, at Children's National Hospital in Washington. U.S. regulators are urging drugmaker Pfizer to apply for emergency authorization for a two-dose regimen of its COVID-19 vaccine for children ages 5 and under while awaiting data on a three-dose course, aiming to clear the way for the shots as soon as late February. (AP Photo/Carolyn Kaster, File)
(AP Photo/Carolyn Kaster, File)
(CNN) — The US Food and Drug Administration announced Friday that it is postponing the meeting of its Vaccines and Related Biological Products Advisory Committee, originally scheduled for February 15, as “new data have recently emerged” regarding Pfizer and BioNTech’s emergency use authorization request for their Covid-19 vaccine for children younger than 5.
“This will give the agency time to consider the additional data, allowing for a transparent public discussion as part of our usual scientific and regulatory processes for COVID-19 vaccines. We will provide an update on timing for the advisory committee meeting once we receive additional data on a third dose in this age group from the company’s ongoing clinical trial and have an opportunity to complete an updated evaluation,” acting FDA Commissioner Dr. Janet Woodcock and Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research, said in a statement.
This is a developing story and will be updated.
The-CNN-Wire
™ & © 2022 Cable News Network, Inc., a WarnerMedia Company. All rights reserved.